Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
Priyanka Theophilus
Delineating the mechanism of aggregate or biofilm formation in the Lyme disease spirochete Borrelia burgdorferi
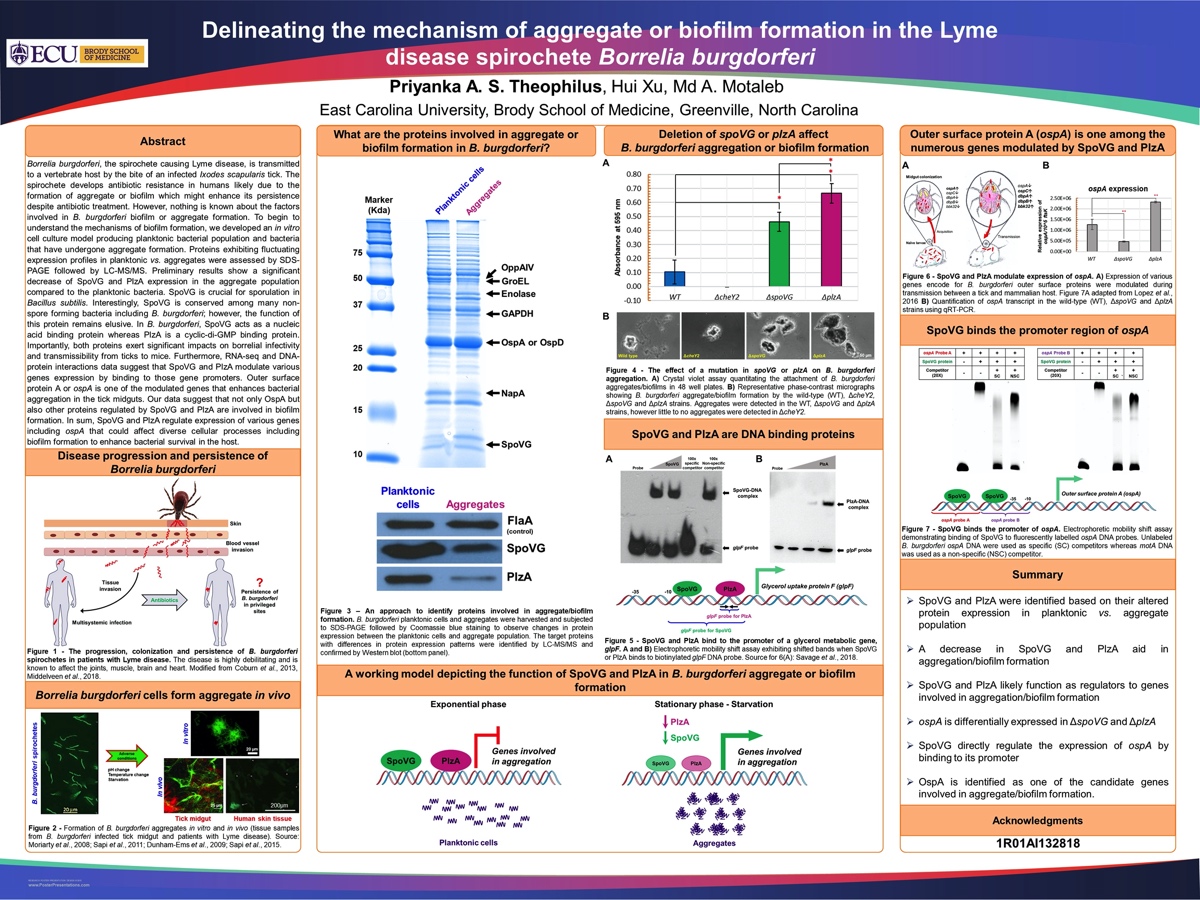
Borrelia burgdorferi, the spirochete causing Lyme disease, is transmitted to a vertebrate host by the bite of an infected Ixodes scapularis tick. The spirochete develops antibiotic resistance in humans likely due to the formation of aggregate or biofilm which might enhance its persistence despite antibiotic treatment. However, nothing is known about the factors involved in B. burgdorferi biofilm or aggregate formation. To begin to understand the mechanisms of biofilm formation, we developed an in vitro cell culture model producing planktonic bacterial population and bacteria that have undergone aggregate formation. Proteins exhibiting fluctuating expression profiles in planktonic vs. aggregates were assessed by SDS-PAGE followed by LC-MS/MS. Preliminary results show a significant decrease of SpoVG and PlzA expression in the aggregate population compared to the planktonic bacteria. SpoVG is crucial for sporulation in Bacillus subtilis. Interestingly, SpoVG is conserved among many non-spore forming bacteria including B. burgdorferi; however, the function of this protein remains elusive. In B. burgdorferi, SpoVG acts as a nucleic acid binding protein whereas PlzA is a cyclic-di-GMP binding protein. Importantly, both proteins exert significant impacts on borrelial infectivity and transmissibility from ticks to mice. Furthermore, RNA-seq and DNA-protein interactions data suggest that SpoVG and PlzA modulate various genes expression by binding to those gene promoters. Outer surface protein A or ospA is one of the modulated genes that enhances bacterial aggregation in the tick midguts. Our data suggest that not only OspA but also other proteins regulated by SpoVG and PlzA are involved in biofilm formation. In sum, SpoVG and PlzA regulate expression of various genes including ospA that could affect diverse cellular processes including biofilm formation to enhance bacterial survival in the host.
Resume / CV
- Resume / CV
Supporting Documents
- Poster
School Work
- Abstract
- Figure 1
- Figure 2
- Figure 3
- Figure 4
- Figure 5
- Figure 6
- Figure 7
- Model
- Summary
References
- Priyanka Theophilus
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
