Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
The PP2A serine/threonine phosphatase complex functions in regulating dendritic morphology
Anukruthi Venukadasula - Shatabdi Bhattacharjee - Dr. Daniel N. Cox
The PP2A Serine/Threonine Phosphatase Complex Functions In Regulating Dendritic Morphology
Anukruthi Venukadasula, Shatabdi Bhattacharjee, Daniel N. Cox
Neuroscience Institute, Georgia State University
Atlanta, GA 30303
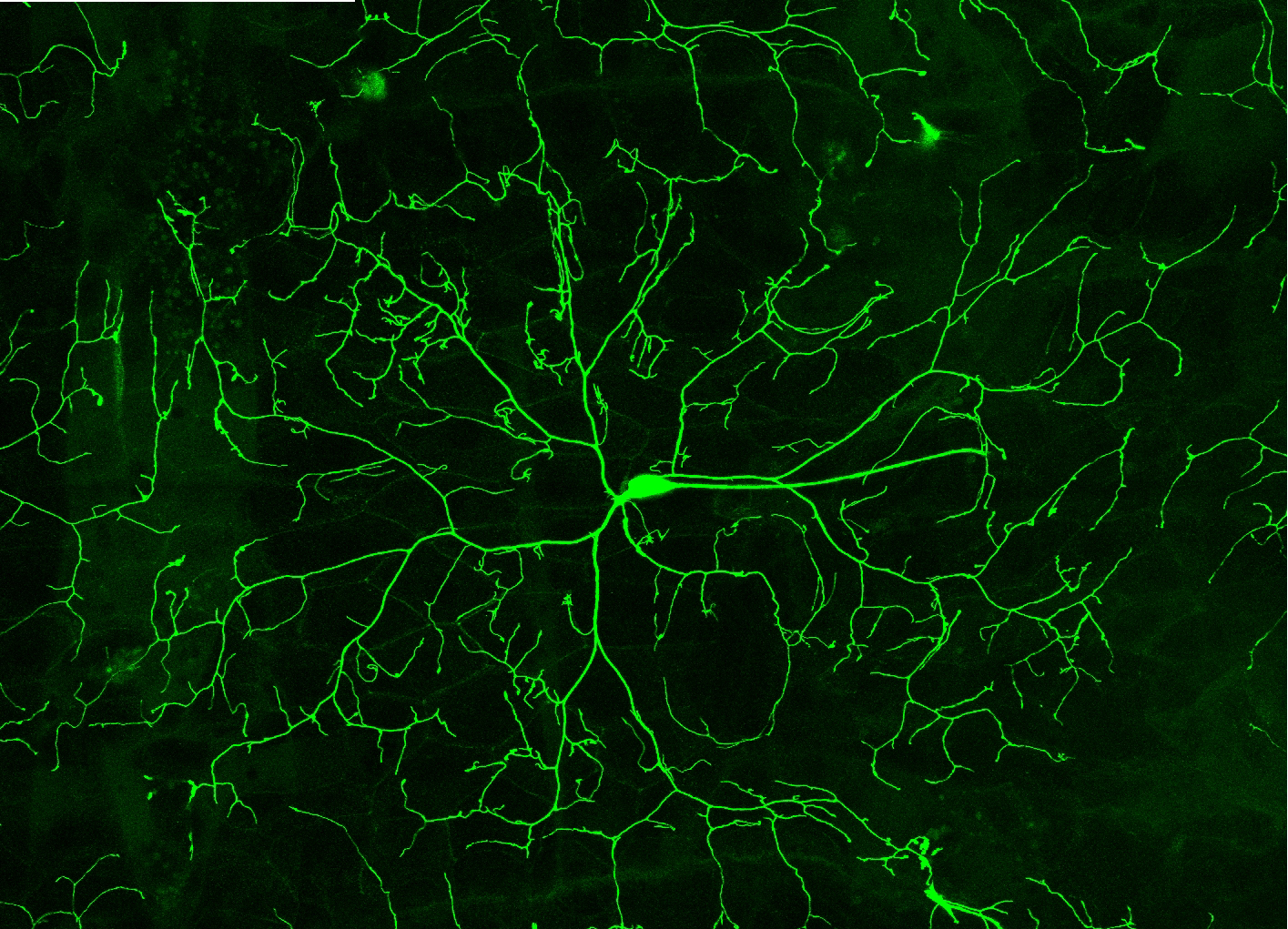
The immense array of morphological diversity exhibited by neurons is critical in specifying patterns of synaptic connectivity. Therefore, discovering the molecular mechanisms that regulate dendritic diversification is essential to understanding the formation and modulation of functional neural circuitry. Previous studies in our lab has shown that the highly conserved PP2A serine/threonine phosphatase complex regulates dendritic diversification in multidendritic (md) neurons in the peripheral nervous system of the fruit fly Drosophila melanogaster. The PP2A complex is composed of a catalytic subunit encoded by microtubule star (mts), a scaffolding subunit encoded by PP2A-29B and one of four alternate regulatory subunits encoded by widerborst (wdb), twins, well-rounded (wrd) and CG4733. Mutant analyses of mts and PP2A-29B reveal severe reductions in dendritic arborization with wdb appearing to function as the relevant regulatory subunit in Class IV (CIV) md sensory neurons. In contrast, mutations in mts and PP2A-29B leads to increased dendritic complexity via de novo filopodia formation in Class I (CI) md sensory neurons. To gain mechanistic insights into the role of PP2A in promoting dendritic arborization, we tried to identify putative targets of the complex in these neurons. Twinstar (Tsr) is an actin severing protein that essential for axonal growth in Drosophila. Phosphorylation of Twinstar by LIMK1 inhibits the protein while its dehosphorylation by the phosphatase Slingshot leads to its activation. Studies in human T lymphocytes have shown that the human homolog of Twinstar, Cofilin, is dephosphrylated by PP2A. Preliminary phenotypic analysis has shown that the knockdown of tsr leads to loss of dendritic complexity in CIV md neurons. In addition, expressing a phosphomimetic form of tsr leads to reduction in both the total dendritic length and branches while expressing a phosphoresistant form of tsr does not affect morphology. Further, expressing the phosphoresistant from of tsr in neurons where mts was knockdown was able to rescue the phenotypic defects. Collectively, these data suggest that Tsr may be a target of PP2A in these neurons.
-
School Work
- Anukruthi Venukadasula - Cox Lab - Neuroscience Institute - GSURC Spring 2021
Media
- GSURC Poster Detailed PPT Presentation
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
