Your message has been sent.
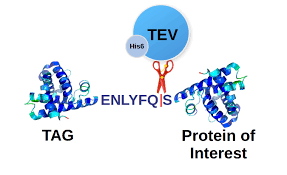
When purified proteins are needed for a project, the best way to obtain a large quantity is in a recombinant form. With complementary DNA (cDNA) and polymerase chain reaction (PCR) methods, the gene coding for the protein of interest can be inserted into the plasmid. After confirming the construct with sequencing, the plasmid can be transformed into the expression host, and single colonies can be used for protein expression. To assist purification of the recombinant protein, an affinity tag may be attached to the protein, followed by a cleavage site, if an affinity chromatography step is needed. If scientists are doing structural or biochemical studies of a protein, the tag needs to be removed because it will interfere with the results. Therefore, a useful molecular tool for protein purification is the tobacco etch virus (TEV) protease that has been widely used for the removal of peptide tags. The purification scheme of TEV protease starts with the transformation of plasmids containing the gene for TEV protease into competent cells, and the bacterium Escherichia coli is one of the frequently used hosts to produce TEV. Then, expression of TEV protease is induced using isopropyl β-D-1-thiogalactopyranoside (IPTG). After lysing the cells to release the proteins, affinity chromatography using a His-tag column is performed so that TEV protease is purified. By knowing how to purify TEV protease, many different approaches can be taken to purify proteins of interest because it is cost-effective to synthesize recombinant proteins that can be cleaved by TEV protease.
-
School Work
- TEV Protease Poster
Media
- Expression and Purification of TEV Protease
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
 CLOSE SIDEBAR
CLOSE SIDEBAR