Your message has been sent.
Calcium (Ca2+) is vital to many biological systems, and it contributes to a variety of processes across different organisms, such as aiding the regulation of ossification. Therefore, it is important to study the mechanisms of how Ca2+ concentrations are monitored in living cells. Measurements of Ca2+ concentration are completed through the calcium-sensing receptor (CaSR). CaSR is a part of the family C of G protein-coupled receptors (GPCRs). The CaSR is apparent in several biological forms and plays a crucial role in the functionality of organisms.
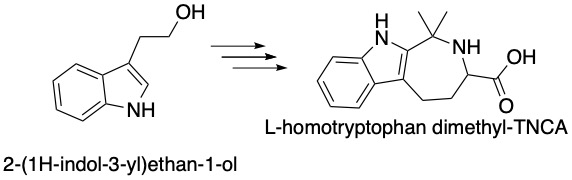
Abnormalities in the CaSR are related to severe diseases, such as autosomal dominant hypocalcemia, so it is important to gain as much knowledge as possible about this receptor. The CaSR functions to dictate the intracellular Ca2+ concentration by reacting to these extracellular stimuli. It is stimulated by the concentration of divalent cations (Ca2+ and Mg2+) and amino acids. Even small changes in the concentration of metal ions can cause large differences in the structure and function of the CaSR. Amino acids can also modulate the functionality of the receptor, usually by acting as co-agonists. A tryptophan derivative, L-1,2,3,4-tetrahydronorharman-3-carboxylic acid (TNCA), can bind to the extracellular domain of CaSR and act as an agonist when paired with metal ions. This poster presentation will outline the current progress in studying the synthesis of TNCA derivatives and its effects on the CaSR.
-
School Work
- Calcium Sensing Receptor and Synthesis of TNCA
Media
- Calcium Sensing Receptor and Synthesis of TNCA Pitch
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
 CLOSE SIDEBAR
CLOSE SIDEBAR