Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
Meeting Number:2624 116 9938
Join by video system
Dial [email protected] and enter your host PIN 4417.
You can also dial 173.243.2.68 and enter your meeting number.
Join by phone
+1-415-655-0002 US Toll
Access code: 2624 116 9938
Host PIN: 4417
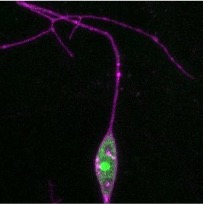
Ribosomal Proteins Have Heterogeneous Effects on Dendritic Morphology in CI md Neurons
EA Timmins-Wilde, S Bhattacharjee, EN Lottes, V Veeranki, DN Cox (Faculty Sponsor: Dr. Dan Cox)
Ribosomes are cellular organelles that assemble proteins through the process of translation. Ribosomopathies, or diseases of the ribosome, are implicated in developmental neurological disorders such as microcephaly. Recent evidence suggests protein synthesis may be regulated not only at the transcriptional level, but by specificity of ribosomal structure. Eukaryote ribosomes (80S) are composed of a large subunit (60S), and a small subunit (40S), altogether consisting of 79 ribosomal proteins (RPs). We knocked down (KD) 30 individual RPs in Class I (CI) multidendritic (md) neurons of the peripheral nervous system in Drosophila melanogaster larvae. KD of 7 RP genes in CI md neurons decreased total dendritic length (tdl) by over 25%, indicating a severe disruption in morphology. 11 RP KD had mild to no effect on dendritic morphology. Interestingly, 2 RP KD increased tdl by over 10%, adding complexity to the arbor. To further look at the effect of RP KD on ribosome localization, we used RPL10Ab::GFP as a selective marker for visualizing ribosomal distribution along the dendritic arbor. Our results showed that in CI neurons, RPL36A and RPL7 KD resulted in almost complete abolition of ribosomal fluorescence along all dendritic branches. Moreover, fluorescence was localized specifically in the nucleus, indicating possible disruption of 40S or 60S subunit assembly. Together, our results provide evidence that KD of different RPs have varying severities of effect on the cell, suggesting the roles of RPs are not homologous.
-
School Work
- Ribosomal proteins have heterogeneous effects on dendritic morphology in CI md neurons
Media
- A brief overview of my research
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy

