Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
Optimizing Purification of Collagen targeted Protein MRI contrast agent for early Detection
Hyein Kang, Brenda-Ruth Mimba, Doungjun Li (Faculty Sponsor: Dr.Jenny Yang)
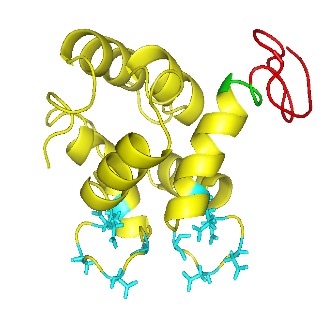
hProCA32. Collagen 1 protein is a newly developed promising MRI contrast agent used to detect small, early stages of liver tumors and other liver diseases like liver fibrosis and cirrhosis. To prepare hProCA32.collagen protein for preclinical and clinical applications and to facilitate drug discoveries, we need to develop an optimized expression and purification process with a high yield. hProCA32.collagen contains two cysteine residues and the formation of intra-disulfide bond is required for its targeting capability. However, there is a challenge to the formation of inter-disulfide bonds, which results in protein aggregation and disulfide bond shuffling. In this study, we first report the expression of hProCA32.collagen using an E.coli expression system with an average yield of cell pellet of 6g/L with desired protein expressed. The goal of the purification was to purify the protein in order to separate hProCA32. Collagen from other unwanted host cell proteins, and refold the protein down to its monomeric form while increasing the protein yield after the purification process. The purification process was optimized by changing the FPLC purification column from Q column to SP column and adjusting buffer pH from 7.4 to pH 5.5 and pH 6.0. As the result, using the FPLC purification with SP column with pH 6.0, dimers, high oligomers, and other impurities were significantly decreased, and the sample mainly consisted of about 80% monomer. The purified protein exhibits high MRI contrast capability with high relaxivity, r1 and r2 that are several folds higher than clinically approved contrasts. Further optimizing this process for purification and formulation will facilitate the early detection of cancer and other diseases by non-invasive MRI.
-
School Work
- Optimizing Purification of Collagen targeted Protein MRI contrast agent for early Detection
Media
- Elevator Pitch
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
