Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
Cp* Iridium Complexes With Phenyliminoquinone-based Ligands
Ashleigh Arrington, Joseph Spiconardi, Miles Wilklow-Marnell (faculty mentor)
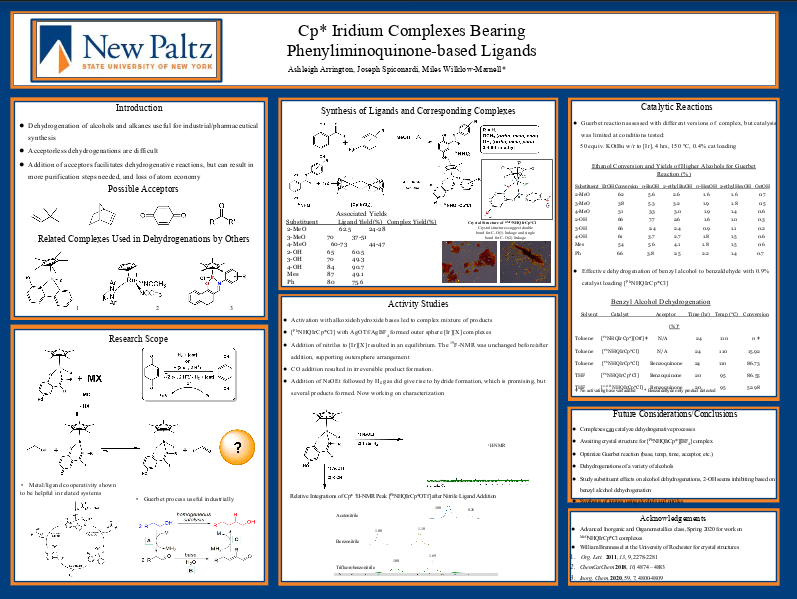
A set of N,O binding, quinone-based ligands including 1,4-dihydroxy-benzylidine-phenylimine (PhNHQ) and other variations with substitutions on the phenyl ring were prepared. Half-sandwich iridium complexes of the general formula PhNHQIrCp*Cl were synthesized using [Cp*IrCl2]2 as the iridium source, and characterized via X-ray diffraction and NMR spectroscopy. The complexes have been assessed as catalysts in several reactions, including the Guerbet upgrading of ethanol to higher alcohols. Coordinatively unsaturated cationic derivatives of the PhNHQ complex were prepared with triflate and tetrafluoroborate counterions by reaction with their corresponding silver salts, and characterized by 1H and 19F-NMR spectroscopy. Ligand addition studies of these complexes has been conducted with a series of nitrile-based ligands, CO, and triphenylphosphine, monitored by 1H-NMR spectroscopy. The formation of iridium hydride complexes by activation of H2 was observed after reaction of [PhNHQIrCp*][Otf] with sodium ethoxide followed by exposure to 1 atm H2.
-
School Work
- NHQIrCp* Complexes
Media
- Elevator Talk for Ashleigh Arrington, Joseph Spiconardi, and Miles Wilklow-Marnell
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
