Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
The Impact of Dynamic Perfusion on Ex-Vivo 3-Dimensional Vasculogenesis: Lessons for the Future
Josh Grosser
Poster Title: The Impact of Dynamic Perfusion on Ex-Vivo 3-Dimensional Vasculogenesis: Lessons for the Future
Student: Joshua Grosser, Class of 2024
Faculty Mentor and Department: Dr. Adam Katz, Department of Plastic and Reconstructive Surgery
Funding Source: Wake Forest Institute of Regenerative Medicine
ABSTRACT
Background: While regenerative medicine has the potential to revolutionize the treatment of tissue defects, nutrient delivery is a major hurdle to growing complex organs. To address this challenge, Dr. Katz has proposed a therapeutic platform which combines principles of perfusion bioreactors, negative pressure wound therapy, and “bottom-up” tissue regeneration (whereby cell-laden “modules” organize into higher order ‘neo-tissues’). Negative pressure dressings, which are already used clinically to assist in would healing, would pull nutrients, growth factors, and other biomodulators through a void space packed with autologous stem cells, keeping them alive and guiding their differentiation. To gauge the ability of cells to grow and self-assemble under these conditions, adipose-derived cells (AdCs) and human umbilical vein endothelial cells (HUVECs) were co-cultured on microbeads and grown in a 3D printed bioreactor.
Hypothesis: Co-cultures grown under dynamic conditions will exhibit greater total cell proliferation than co-cultures grown under static conditions. This difference will be most apparent in the middle layer of the microbead packed mesh.
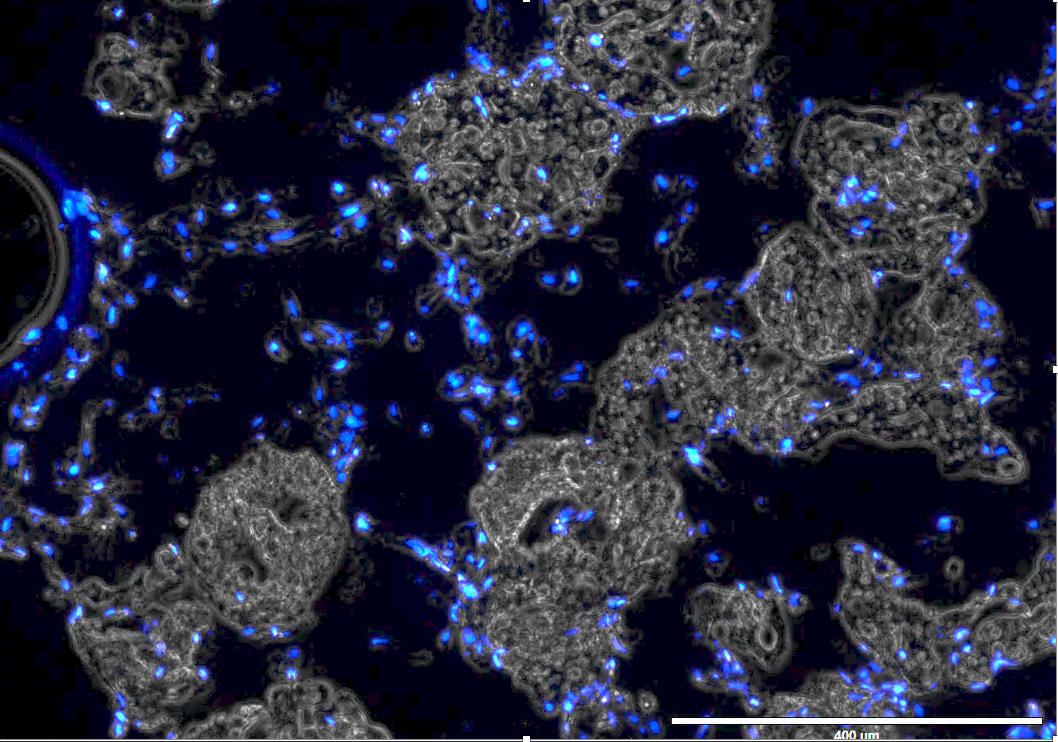
Methods: HUVECs (9.6 million) and AdCs (2.4 million) were combined with 0.25g Cultisphere gelatin microbeads in a low adhesion dish, rocked for 3 days at 37°C and 5% CO2 with media changes daily, and placed in a 40 µm mesh. The mesh was then placed in either a 50 mL conical (“static”) or the bioreactor (“dynamic”). Cells received 24 mL of fresh media daily in both conditions. Cells were harvested, frozen in OCT, sectioned at 16 µm, and DAPI stained at 0 and 2 weeks. Average DAPI+ cells per bead were compared using unpaired t-tests.
Results: There were an average of 17.36 ± 2.21 (SE) cells/bead at day 0. After 2 weeks, cells per bead in the top, middle, and bottom layers of the microbead section were 9.31 ± 1.19, 12.32 ± 1.58, and 10.5 ± 1.48 (static), and 3.71 ± 0.43, 2.63 ± 0.35, and 2.05 ± 0.29 (dynamic), respectively. The average bead diameter was 172.2 ± 8.5 µm and did not differ significantly between groups. The top layer of the dynamic experiment had significantly more cells/bead than the bottom layer (p<0.05), but cell densities did not otherwise differ within groups. All layers within the static and dynamic groups had statistically fewer cells than the week 0 control (p<0.05), except for the middle layer of the static experiment (p=0.07).
Conclusions: Cell density was greatest at the day zero control, indicating that the bioreactor was unsuccessful in facilitating cell proliferation. Cells did not grow better in the middle layer of the dynamic experiment, nor did they grow worse in the middle layer of the static experiment (relative to the outer layers). Lack of oxygen exchange might explain these findings (both conditions were closed to atmosphere), and future bioreactors should draw from a media jar exposed to filtered air. The bioreactor chamber diameter should also be reduced to match the diameter of the microbead-filled mesh, since space around the mesh in the dynamic experiment allowed media to flow around the microbeads as opposed to through them. Alternatively, beads may be cultured directly in the bioreactor well (without the mesh). Lastly, leaking from the sealed bioreactor chamber implied that pressures within the chamber were significant at times. Rather than using a sealed pump/vacuum system to simultaneously push/pull media through the bioreactor, preliminary experiments suggest that an IV pump, set at a constant infusion rate and connected downstream of the bioreactor, can be placed below the chamber, allowing gravity to pull media through the bioreactor at a constant rate without creating extreme pressures.
Source of mentor’s funding or other support that funded this research: Department of Plastic Surgery
-
Supporting Documents
- The Impact of Dynamic Perfusion on Ex-Vivo 3-Dimensional Vasculogenesis: Lessons for the Future
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
