Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
Evaluating Long-term Outcome Trajectories of Selective Laser Amygdalohippocampectomy for Medically Intractable Mesial Temporal Lobe Epilepsy
Alexander C. Horn
Poster Title: Evaluating Long-term Outcome Trajectories of Selective Laser Amygdalohippocampectomy for Medically Intractable Mesial Temporal Lobe Epilepsy
Student: Alexander C. Horn, Class of 2024
Faculty Mentor and Department: Heidi M. Munger Clary, MD, MPH, FACNS
Funding Source: The Harry O. Parker Neuroscience Research Fund
ABSTRACT
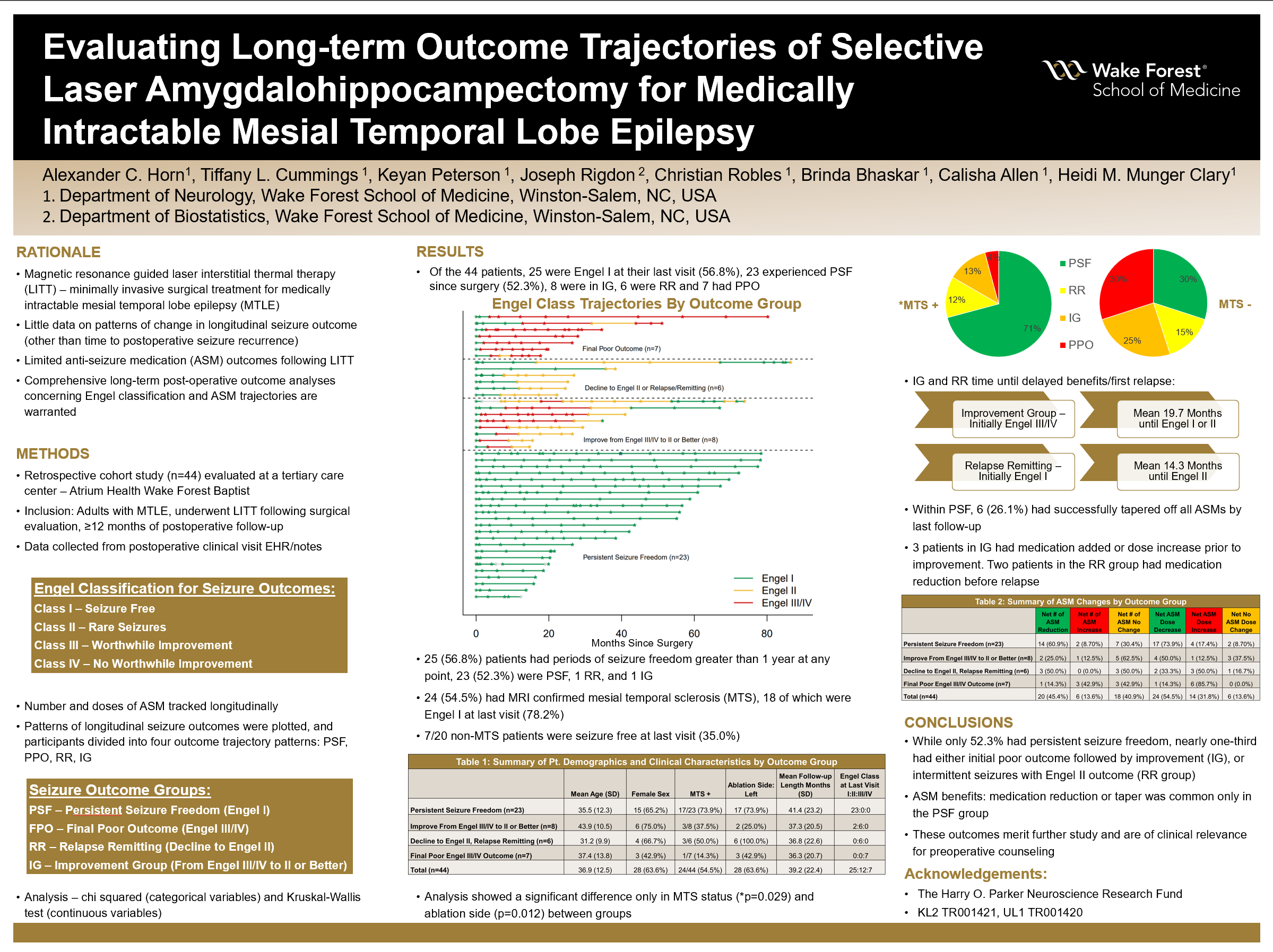
Background: Magnetic resonance guided laser interstitial thermal therapy (LITT), also called laser ablation, is a novel, minimally invasive surgical treatment for medically intractable mesial temporal lobe epilepsy. There is a paucity of data on patterns of change in longitudinal seizure outcome (other than time to postoperative seizure recurrence) and limited to no data examining anti-seizure medication (ASM) outcomes following LITT. Thus, comprehensive long-term post-operative outcome analyses concerning Engel classification and ASM trajectories are warranted.
Hypothesis: (1) With respect to seizure frequency, we expected a portion of those who achieved Engel Class I to have experienced a period of delay or worsening following surgical intervention. Alternatively, we also expect to find patients who purely regress to worse Engel Classifications following periods of seizure freedom. (2) As for AED usage, we anticipated trajectories that reflect a progression towards drug independence, especially in patients who achieve Engel Class I.
Methods: This study utilized retrospective data from 44 patients evaluated by the Wake Forest Baptist Health Comprehensive Epilepsy Center, spanning the past decade. Qualifying patients had medically intractable mesial temporal lobe epilepsy (MTLE) and underwent mesial temporal laser ablation following surgical evaluation. At least 12 months of postoperative follow-up was required for inclusion. Seizure outcomes and ASM data were collected from clinical visits and entered into a REDCap database. Patterns of longitudinal seizure outcomes were plotted, and participants divided into four outcome trajectory patterns: Persistent Seizure Freedom (PSF – continuous Engel I), Persistent Poor Outcome (PPO – continuous Engel III/IV), Relapse Remitting (RR – decline to Engel II), Improvement Group (IG - from Engel III/IV to Engel II or Better).
Results: Patient demographics featured a mean age at surgery of 36.9 years, 28/44 (63.6%) female, 28/44 (63.6%) left sided surgery, and a mean total follow-up length of 39.2 months. Of the 44 patients, 25 were Engel I at their last visit (56.8%), 23 experienced PSF since surgery (52.3%), 8 were IG, 6 were RR and 7 PPO. Twenty-five patients had periods of seizure freedom greater than 1 year at any point, 23 of which were PSF, 1 in the RR group, and 1 in the improvement group. Twenty-four had MRI confirmed mesial temporal sclerosis (MTS), 18 of which were Engel I at their last visit (78.2%). Seven of the 20 non-MTS patients were seizure free at their last visit (35.0%). Those in IG had mean 19.7 months until said delayed benefits, and those in the RR group had mean 14.3 months until first relapse. Within PSF, 6 (26.1%) had successfully tapered off all ASMs by their last follow-up. No patients in the other groups were ASM free. Twenty-four out of 44 patients (54.5%) were taking lower ASM doses at last visit compared to surgery, 17 of which were in the PSF group, 4 in IG, 2 in RR, and 1 in PPO. Three patients in the improvement group had medication added or dose increase prior to improvement. Conversely, 2 patients in the RR group had medication reduction before relapse.
Conclusions: In this long-term follow up study of mesial temporal laser ablation, while only 52.3% had persistent seizure freedom, nearly one-third had either initial poor outcome followed by improvement (IG), or intermittent seizures with Engel II outcome (RR). Medication reduction or taper was common only in the PSF group. These outcomes merit further study and are of clinical relevance for preoperative counseling.
Source of mentor’s funding or other support that funded this research: Wake Forest Baptist Health Department of Neurology
-
Supporting Documents
- Evaluating Long-term Outcome Trajectories of Selective Laser Amygdalohippocampectomy for Medically Intractable Mesial...
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
