Your message has been sent.
 CLOSE SIDEBAR
CLOSE SIDEBAR
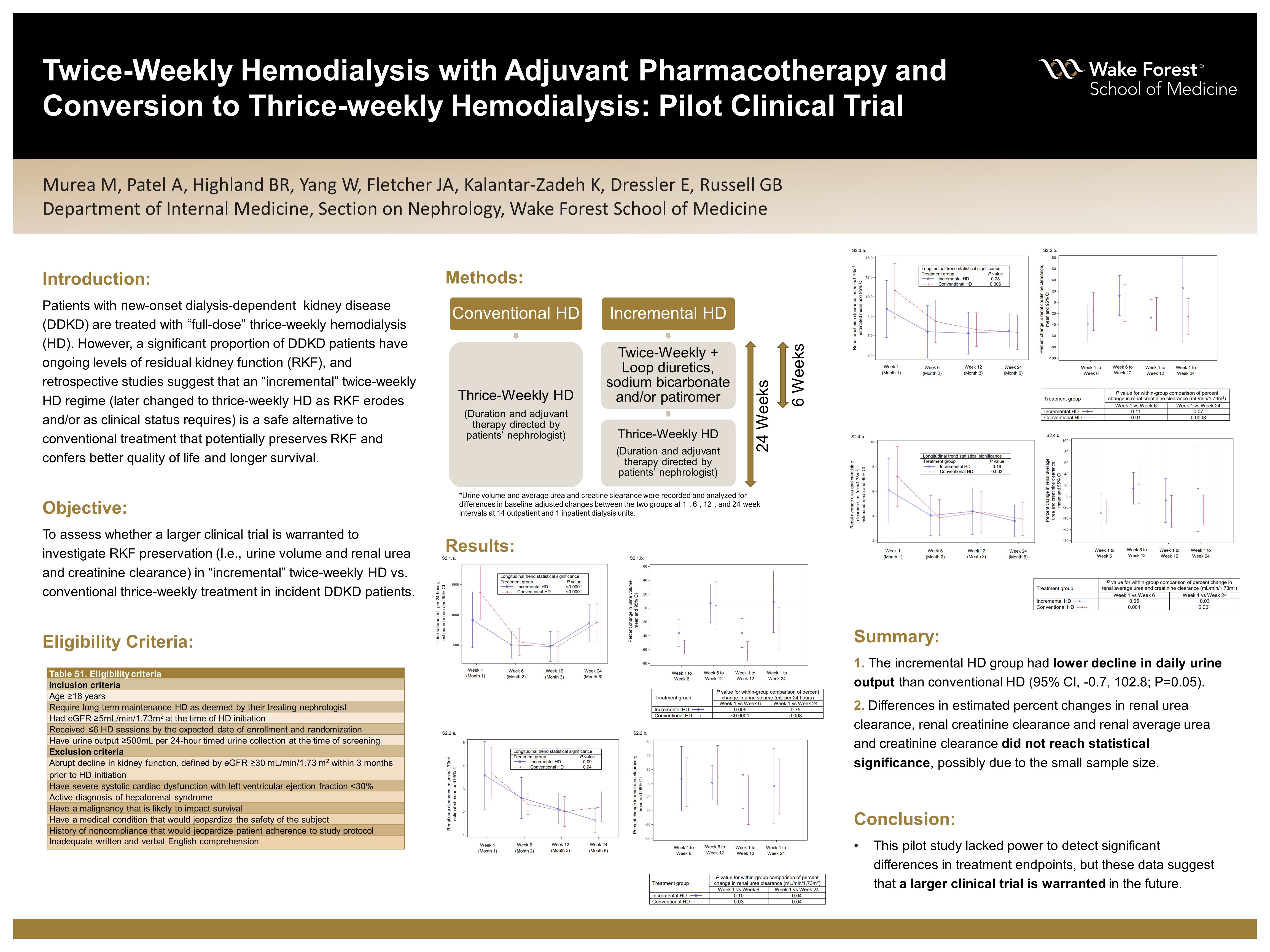
Twice-Weekly Hemodialysis with Adjuvant Pharmacotherapy and Conversion to Thrice-weekly Hemodialysis: A Pragmatic, Fully-Embedded, Individually-Randomized Pilot Clinical Trial
Benjamin Highland
Background: Patients with new-onset dialysis-dependent kidney disease (DDKD) are treated identically, with “full-dose” thrice-weekly HD. Thus, the conventional approach to thrice-weekly HD disregards the heterogeneous nature of DDKD, as a significant proportion of patients have ongoing levels of residual kidney function (RKF) which can complement an initial therapy of less intensive HD. Retrospective studies suggested incremental HD (initial twice-weekly HD, later changed to thrice-weekly HD as RKF erodes and/or as clinical status requires) is a safe alternative to conventional HD with potential benefit longer preservation of RKF which, in turn, can confer better quality of life and longer survival. A pilot clinical trial was conducted to assess the trajectory of RKF parameters between patients treated with incremental-start HD and conventional HD.
Hypothesis: Incremental-start HD (twice-weekly HD for six weeks followed by thrice-weekly HD) will result in better preservation of RKF compared to conventional HD (outright thrice-weekly HD).
Methods: This was an individually randomized, open-label parallel-group pilot controlled trial involving patients with new-onset DDKD who have been on HD for ≤2 weeks, with pre-dialysis baseline estimated glomerular filtration rate ≥5 mL/min/1.73m2 and urine volume ≥500 mL/day. Eligible participants were randomly assigned (1:1 ratio) to twice-weekly HD and adjuvant pharmacologic therapy (loop diuretics, sodium bicarbonate and/or patiromer) for 6 weeks followed by thrice-weekly HD (incremental HD group) or continued thrice-weekly HD (conventional HD group) at 14 outpatient dialysis units and 1 inpatient dialysis unit. Urine output (mL per 24 hours), and average urea and creatinine clearance (mL/min/1.73m2) were recorded and analyzed for differences in baseline-adjusted changes between the two treatment groups.
Results:48 patients were enrolled and randomized to incremental HD (n=23) vs conventional HD (n=25). All participants adhered to timed inter-dialytic urine collections performed at baseline, week 6, week 12 and week 24. At week 24, the between-group difference in percent change of estimated means showed 51.0 percentage points (95% CI, -0.7, 102.8; P=0.05) lower decline in urine volume (mL/day) and 57.9 percentage points (95% CI, -22.6, 138.4; P=0.15) lower decline in renal average urea and creatinine clearance (mL/min/1.73m2) in the incremental HD group. Changes in renal urea clearance did not differ between the groups while renal creatinine clearance had a lower decline in incremental HD group compared to conventional HD group.
Conclusions: The pilot trial suggests better preservation of certain components of RKF with incremental-start HD. Larger clinical trials are warranted to delineate the effect of different HD therapy models on RKF, clinical outcomes and patient-reported outcomes.
Powered by Acadiate
© 2011-2024, Acadiate Inc. or its affiliates · Privacy
